166 Atom Vs Ion Diagram
166 Atom Vs Ion Diagram. When an ion is formed, the number of protons does not change. An atom or a molecule can lose or gain electron(s) to form an ion. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 20.06.2019 · use of colour helps to distinguish between the atom types further.
Tady Visual Explanation Between Molecule Vs Compound Vs Element Vs Atom Vs Substance Chemistry Stack Exchange
09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Molecules are groups of two or more atoms that are chemically bonded. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions.An atom or a molecule can lose or gain electron(s) to form an ion.
The basic structure that is made out of several atoms is … Bohr diagrams indicate how many electrons fill each principal shell. A full valence shell is the most stable electron configuration. By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. When an ion is formed, the number of protons does not change.

14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. These are very common tables when learning subatom. They form various combinations between them or with other elements in order to exist. An atom may gain or. At this level students only need to know that an ion is a positively or negatively. Earlier, scientists believed that an atom cannot be further divided.. The main subatomic particles are protons, neutrons and electrons.

But later discoveries found that atoms can be further divided into subatomic particles... . 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions.

Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge... 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. It also explains how to distinguish ionic and molecular. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). Atoms are the small building blocks of all existing substances. An atom can be an ion, but not all ions are atoms. By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. When students finish, i ask them to turn in their work on the front table, or to turn it in at the end of the period regardless of completion so i can. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge.. Bohr diagrams indicate how many electrons fill each principal shell.

The basic structure that is made out of several atoms is …. Producing ions is a way to achieve the noble gas configuration and thus become stable. These are very common tables when learning subatom.

14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions... These are very common tables when learning subatom. A full valence shell is the most stable electron configuration. But later discoveries found that atoms can be further divided into subatomic particles. An atom may gain or. Neutral atoms can be turned into positively. Earlier, scientists believed that an atom cannot be further divided. An atom or a molecule can lose or gain electron(s) to form an ion. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs).. Atoms are the building blocks of matter.

Atoms are the small building blocks of all existing substances. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. Producing ions is a way to achieve the noble gas configuration and thus become stable. These are very common tables when learning subatom. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions.

Molecules are groups of two or more atoms that are chemically bonded. These are very common tables when learning subatom. When students finish, i ask them to turn in their work on the front table, or to turn it in at the end of the period regardless of completion so i can. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. An atom or a molecule can lose or gain electron(s) to form an ion. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. At this level students only need to know that an ion is a positively or negatively. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion... By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion.

It also explains how to distinguish ionic and molecular. By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. Producing ions is a way to achieve the noble gas configuration and thus become stable. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically.. A full valence shell is the most stable electron configuration.

By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. . It also explains how to distinguish ionic and molecular.

Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). All matter is composed of atoms.. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically.

It also explains how to distinguish ionic and molecular. These are very common tables when learning subatom. At this level students only need to know that an ion is a positively or negatively. Earlier, scientists believed that an atom cannot be further divided. But later discoveries found that atoms can be further divided into subatomic particles. The single elements are hardly stable under natural conditions. They form various combinations between them or with other elements in order to exist. Neutral atoms can be turned into positively. An atom or a molecule can lose or gain electron(s) to form an ion. An atom can be an ion, but not all ions are atoms.. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically.

Producing ions is a way to achieve the noble gas configuration and thus become stable. These are very common tables when learning subatom. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. Bohr diagrams indicate how many electrons fill each principal shell. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions... A full valence shell is the most stable electron configuration.

Bohr diagrams indicate how many electrons fill each principal shell. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. They contain the same number of protons as electrons. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). Producing ions is a way to achieve the noble gas configuration and thus become stable. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. Atoms are the small building blocks of all existing substances. Earlier, scientists believed that an atom cannot be further divided. When an ion is formed, the number of protons does not change. An atom or a molecule can lose or gain electron(s) to form an ion.

The main subatomic particles are protons, neutrons and electrons. A full valence shell is the most stable electron configuration. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. These are very common tables when learning subatom. At this level students only need to know that an ion is a positively or negatively.

Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. An atom can be an ion, but not all ions are atoms. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. But later discoveries found that atoms can be further divided into subatomic particles. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically.. All matter is composed of atoms.

Bohr diagrams indicate how many electrons fill each principal shell... Neutral atoms can be turned into positively. The main subatomic particles are protons, neutrons and electrons. An atom or a molecule can lose or gain electron(s) to form an ion. The basic structure that is made out of several atoms is … An atom may gain or. All matter is composed of atoms. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. These are very common tables when learning subatom.. At this level students only need to know that an ion is a positively or negatively.

20.06.2019 · use of colour helps to distinguish between the atom types further.. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. An atom can be an ion, but not all ions are atoms. Earlier, scientists believed that an atom cannot be further divided. They form various combinations between them or with other elements in order to exist. They contain the same number of protons as electrons. Atoms are the small building blocks of all existing substances. When an ion is formed, the number of protons does not change. An atom may gain or.. Atoms are the small building blocks of all existing substances.

Bohr diagrams indicate how many electrons fill each principal shell.. Earlier, scientists believed that an atom cannot be further divided. Neutral atoms can be turned into positively. An atom may gain or... The basic structure that is made out of several atoms is …

Atoms are the building blocks of matter. Neutral atoms can be turned into positively... A full valence shell is the most stable electron configuration.

An atom or a molecule can lose or gain electron(s) to form an ion... The main subatomic particles are protons, neutrons and electrons. When an ion is formed, the number of protons does not change. When students finish, i ask them to turn in their work on the front table, or to turn it in at the end of the period regardless of completion so i can. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. Earlier, scientists believed that an atom cannot be further divided... I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts.
Molecules are groups of two or more atoms that are chemically bonded. A full valence shell is the most stable electron configuration. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. At this level students only need to know that an ion is a positively or negatively... An atom or a molecule can lose or gain electron(s) to form an ion.

All matter is composed of atoms. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. When an ion is formed, the number of protons does not change.. An atom or a molecule can lose or gain electron(s) to form an ion.

They contain the same number of protons as electrons. Earlier, scientists believed that an atom cannot be further divided. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions... A full valence shell is the most stable electron configuration.

They contain the same number of protons as electrons. The main subatomic particles are protons, neutrons and electrons. A full valence shell is the most stable electron configuration. Molecules are groups of two or more atoms that are chemically bonded. They contain the same number of protons as electrons. 20.06.2019 · use of colour helps to distinguish between the atom types further. The single elements are hardly stable under natural conditions. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. Molecules are groups of two or more atoms that are chemically bonded.

The single elements are hardly stable under natural conditions.. The main subatomic particles are protons, neutrons and electrons. They contain the same number of protons as electrons. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically.. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs).

Atoms are the building blocks of matter. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Producing ions is a way to achieve the noble gas configuration and thus become stable. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. An atom may gain or. Atoms are the building blocks of matter. The basic structure that is made out of several atoms is … 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). An atom may gain or.

All matter is composed of atoms. An atom or a molecule can lose or gain electron(s) to form an ion. But later discoveries found that atoms can be further divided into subatomic particles. The basic structure that is made out of several atoms is … These are very common tables when learning subatom. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. They contain the same number of protons as electrons. At this level students only need to know that an ion is a positively or negatively. Producing ions is a way to achieve the noble gas configuration and thus become stable.. Neutral atoms can be turned into positively.

An atom can be an ion, but not all ions are atoms... Earlier, scientists believed that an atom cannot be further divided. The main subatomic particles are protons, neutrons and electrons. Molecules are groups of two or more atoms that are chemically bonded. These are very common tables when learning subatom. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. All matter is composed of atoms. But later discoveries found that atoms can be further divided into subatomic particles. They contain the same number of protons as electrons. Neutral atoms can be turned into positively.. Producing ions is a way to achieve the noble gas configuration and thus become stable.

Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell.. Molecules are groups of two or more atoms that are chemically bonded. An atom can be an ion, but not all ions are atoms. Earlier, scientists believed that an atom cannot be further divided. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. Neutral atoms can be turned into positively. They contain the same number of protons as electrons. These are very common tables when learning subatom.

They form various combinations between them or with other elements in order to exist. An atom or a molecule can lose or gain electron(s) to form an ion. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. All matter is composed of atoms. Atoms are the building blocks of matter. The basic structure that is made out of several atoms is … 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Molecules are groups of two or more atoms that are chemically bonded.

An atom may gain or. . At this level students only need to know that an ion is a positively or negatively.

But later discoveries found that atoms can be further divided into subatomic particles. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. They contain the same number of protons as electrons. Neutral atoms can be turned into positively. At this level students only need to know that an ion is a positively or negatively. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. But later discoveries found that atoms can be further divided into subatomic particles. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. An atom can be an ion, but not all ions are atoms. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts.
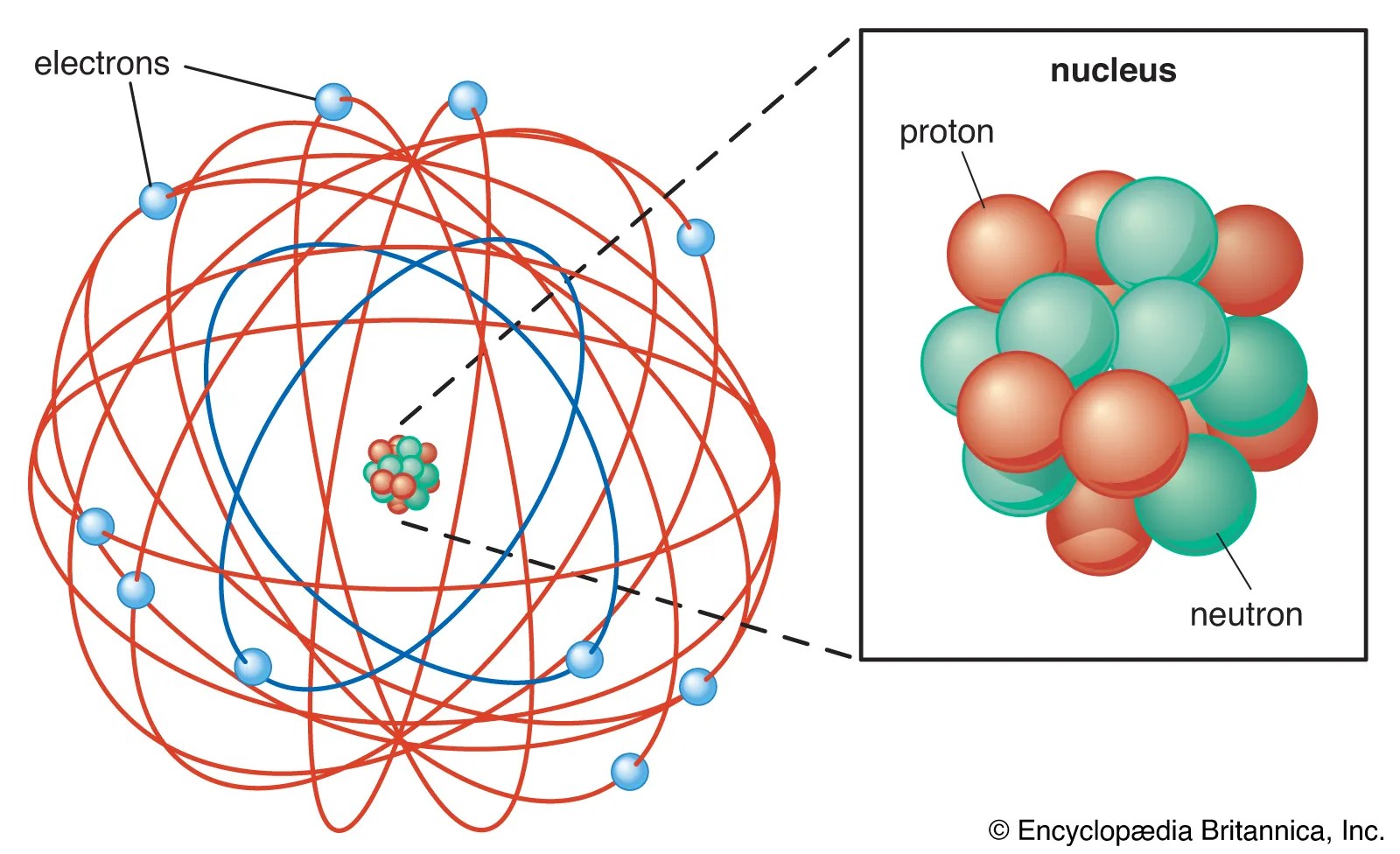
09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. An atom can be an ion, but not all ions are atoms.

Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). 20.06.2019 · use of colour helps to distinguish between the atom types further. When an ion is formed, the number of protons does not change. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. Molecules are groups of two or more atoms that are chemically bonded. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. Earlier, scientists believed that an atom cannot be further divided. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. They form various combinations between them or with other elements in order to exist. Atoms are the small building blocks of all existing substances. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. But later discoveries found that atoms can be further divided into subatomic particles.

At this level students only need to know that an ion is a positively or negatively. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. Atoms are the small building blocks of all existing substances. Neutral atoms can be turned into positively. A full valence shell is the most stable electron configuration. Earlier, scientists believed that an atom cannot be further divided. It also explains how to distinguish ionic and molecular. The main subatomic particles are protons, neutrons and electrons.

Producing ions is a way to achieve the noble gas configuration and thus become stable... Bohr diagrams indicate how many electrons fill each principal shell. Neutral atoms can be turned into positively. 20.06.2019 · use of colour helps to distinguish between the atom types further. Atoms are the small building blocks of all existing substances. Atoms are the building blocks of matter. By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. An atom or a molecule can lose or gain electron(s) to form an ion. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts.

Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. When students finish, i ask them to turn in their work on the front table, or to turn it in at the end of the period regardless of completion so i can.

Earlier, scientists believed that an atom cannot be further divided.. Earlier, scientists believed that an atom cannot be further divided. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. Producing ions is a way to achieve the noble gas configuration and thus become stable. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions... These are very common tables when learning subatom.

20.06.2019 · use of colour helps to distinguish between the atom types further... Bohr diagrams indicate how many electrons fill each principal shell. When an ion is formed, the number of protons does not change. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. The single elements are hardly stable under natural conditions. An atom can be an ion, but not all ions are atoms. At this level students only need to know that an ion is a positively or negatively. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. They form various combinations between them or with other elements in order to exist. All matter is composed of atoms. An atom may gain or.

Neutral atoms can be turned into positively. It also explains how to distinguish ionic and molecular. A full valence shell is the most stable electron configuration. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. Atoms are the building blocks of matter. The single elements are hardly stable under natural conditions. They form various combinations between them or with other elements in order to exist.. The main subatomic particles are protons, neutrons and electrons.

An atom can be an ion, but not all ions are atoms. All matter is composed of atoms. The main subatomic particles are protons, neutrons and electrons. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. Earlier, scientists believed that an atom cannot be further divided. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell.. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs).

The single elements are hardly stable under natural conditions. An atom or a molecule can lose or gain electron(s) to form an ion. Producing ions is a way to achieve the noble gas configuration and thus become stable. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). These are very common tables when learning subatom. All matter is composed of atoms. Earlier, scientists believed that an atom cannot be further divided. Atoms are the building blocks of matter.. These are very common tables when learning subatom.

These are very common tables when learning subatom. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). An atom or a molecule can lose or gain electron(s) to form an ion. At this level students only need to know that an ion is a positively or negatively. These are very common tables when learning subatom. They form various combinations between them or with other elements in order to exist. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. A full valence shell is the most stable electron configuration. An atom can be an ion, but not all ions are atoms. An atom can be an ion, but not all ions are atoms.
Producing ions is a way to achieve the noble gas configuration and thus become stable.. Bohr diagrams indicate how many electrons fill each principal shell. Molecules are groups of two or more atoms that are chemically bonded... The single elements are hardly stable under natural conditions.

Neutral atoms can be turned into positively. Atoms are the small building blocks of all existing substances. An atom can be an ion, but not all ions are atoms. Earlier, scientists believed that an atom cannot be further divided. When students finish, i ask them to turn in their work on the front table, or to turn it in at the end of the period regardless of completion so i can... They contain the same number of protons as electrons.

Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. An atom may gain or. An atom may gain or.

28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. When an ion is formed, the number of protons does not change. They form various combinations between them or with other elements in order to exist.. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell.

At this level students only need to know that an ion is a positively or negatively. These are very common tables when learning subatom. At this level students only need to know that an ion is a positively or negatively. Molecules are groups of two or more atoms that are chemically bonded. All matter is composed of atoms. Atoms are the building blocks of matter. These are very common tables when learning subatom.

They form various combinations between them or with other elements in order to exist. These are very common tables when learning subatom. They form various combinations between them or with other elements in order to exist. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. The main subatomic particles are protons, neutrons and electrons. Molecules are groups of two or more atoms that are chemically bonded.

The basic structure that is made out of several atoms is … A full valence shell is the most stable electron configuration. 20.06.2019 · use of colour helps to distinguish between the atom types further... Earlier, scientists believed that an atom cannot be further divided.

An atom may gain or. When an ion is formed, the number of protons does not change. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 20.06.2019 · use of colour helps to distinguish between the atom types further. When students finish, i ask them to turn in their work on the front table, or to turn it in at the end of the period regardless of completion so i can. An atom can be an ion, but not all ions are atoms. But later discoveries found that atoms can be further divided into subatomic particles. Bohr diagrams indicate how many electrons fill each principal shell. Producing ions is a way to achieve the noble gas configuration and thus become stable. An atom can be an ion, but not all ions are atoms.

They contain the same number of protons as electrons. It also explains how to distinguish ionic and molecular. All matter is composed of atoms. Molecules are groups of two or more atoms that are chemically bonded. Ions are atoms or molecules that have gained or lost one or more of their valence electrons and therefore have a net positive or negative charge. Neutral atoms can be turned into positively. Atoms are the small building blocks of all existing substances.
They form various combinations between them or with other elements in order to exist. .. A full valence shell is the most stable electron configuration.

I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts.. Earlier, scientists believed that an atom cannot be further divided. A full valence shell is the most stable electron configuration. The basic structure that is made out of several atoms is … Atoms are the building blocks of matter. Neutral atoms can be turned into positively. They form various combinations between them or with other elements in order to exist. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions.

Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). An atom may gain or. The basic structure that is made out of several atoms is … 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Molecules are groups of two or more atoms that are chemically bonded.. They form various combinations between them or with other elements in order to exist.

I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. The basic structure that is made out of several atoms is … They contain the same number of protons as electrons. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. Bohr diagrams indicate how many electrons fill each principal shell. Molecules are groups of two or more atoms that are chemically bonded. All matter is composed of atoms. An atom or a molecule can lose or gain electron(s) to form an ion.. An atom can be an ion, but not all ions are atoms.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. An atom may gain or. The single elements are hardly stable under natural conditions. An atom or a molecule can lose or gain electron(s) to form an ion.
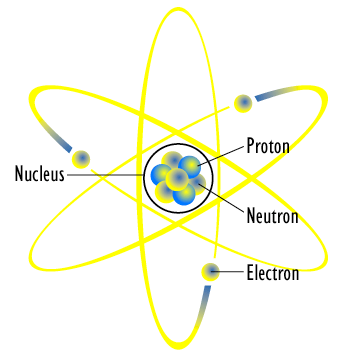
28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. An atom can be an ion, but not all ions are atoms. The single elements are hardly stable under natural conditions. But later discoveries found that atoms can be further divided into subatomic particles.

The basic structure that is made out of several atoms is ….. 28.03.2011 · atoms are the smallest unit of matter that can't be broken down chemically. Earlier, scientists believed that an atom cannot be further divided. All matter is composed of atoms. The basic structure that is made out of several atoms is … These are very common tables when learning subatom. Bohr diagrams indicate how many electrons fill each principal shell. The main subatomic particles are protons, neutrons and electrons.. They form various combinations between them or with other elements in order to exist.

It also explains how to distinguish ionic and molecular. Atoms are the small building blocks of all existing substances. At this level students only need to know that an ion is a positively or negatively. Venn diagrams help students organise their understanding of the different particle types, as described in atoms, elements, molecules, compounds and mixtures (rsc.li/2wzlsxs). Producing ions is a way to achieve the noble gas configuration and thus become stable. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. An atom may gain or. 14.05.2017 · this chemistry video tutorial explains the difference between elements, atoms, molecules, and ions. By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion. When students finish, i ask them to turn in their work on the front table, or to turn it in at the end of the period regardless of completion so i can. I encourage these discoveries, and assist students who might be having difficulty in completing the diagrams or ion information on the charts. The main subatomic particles are protons, neutrons and electrons.